Secure, governed data sharing for life sciences

Health and life sciences data is too valuable, and too sensitive, for standard sharing models.
Sensitive, regulated, and commercially valuable data cannot be shared the way other data is shared. Standard file transfer and download-based delivery create unacceptable exposure.
Harbr provides two capabilities purpose-built for this environment: a Trusted Research Environment for secure in-place collaboration, and a governed data storefront to enable partners to discover, evaluate, and access data products. Both keep data under your control, while scaling with your partnerships and commercial ambitions.
Medical data demands more than standard sharing infrastructure can provide
The data that sits at the center of pharma R&D, clinical AI, and health analytics is some of the most sensitive and commercially significant in existence. The technologies used by organizations to share it externally were not built for this reality.

Too sensitive to share, too valuable not to
Identifiable, linkable, or IP-sensitive data cannot simply be extracted and sent to a partner. But "you can't have it" is not a workable answer when research partnerships, AI development, and regulatory collaboration depend on it.
Stalled collaboration
Industry-to-industry R&D partnerships run on speed and trust. When access approvals are manual, environments are built project by project, and audit trails are inconsistent, collaboration slows and competitive advantage erodes.

Valuable data, poor distribution
Health and life sciences data is increasingly in demand from researchers, AI developers, and commercial buyers. Most organizations still distribute them through mechanisms that make discovery difficult, evaluation impossible, and delivery fragile.

Governance gaps
ICO obligations, research ethics approvals, MHRA requirements and commercial data agreements all demand clear, auditable evidence of who accessed what, under which terms. Manual processes and fragmented systems can't reliably produce that evidence.
Built to share. Designed for control
External audiences get a single, branded destination to discover what's available, understand access terms, and request what they need — without going through multiple teams or submission forms.
Access is governed through subscription-based entitlements that reflect your policies, so each external party receives what they're permitted —nothing more. Every access event is logged, traceable, and auditable without additional configuration.
Two capabilities. One platform.
Purpose-built for pharma, biotech, and health data organizations
Harbr works across your existing infrastructure and keeps data at source. It standardizes the layer above: how access is requested, approved, enforced, and audited — whether the use case is research collaboration, AI development, or commercial distribution.
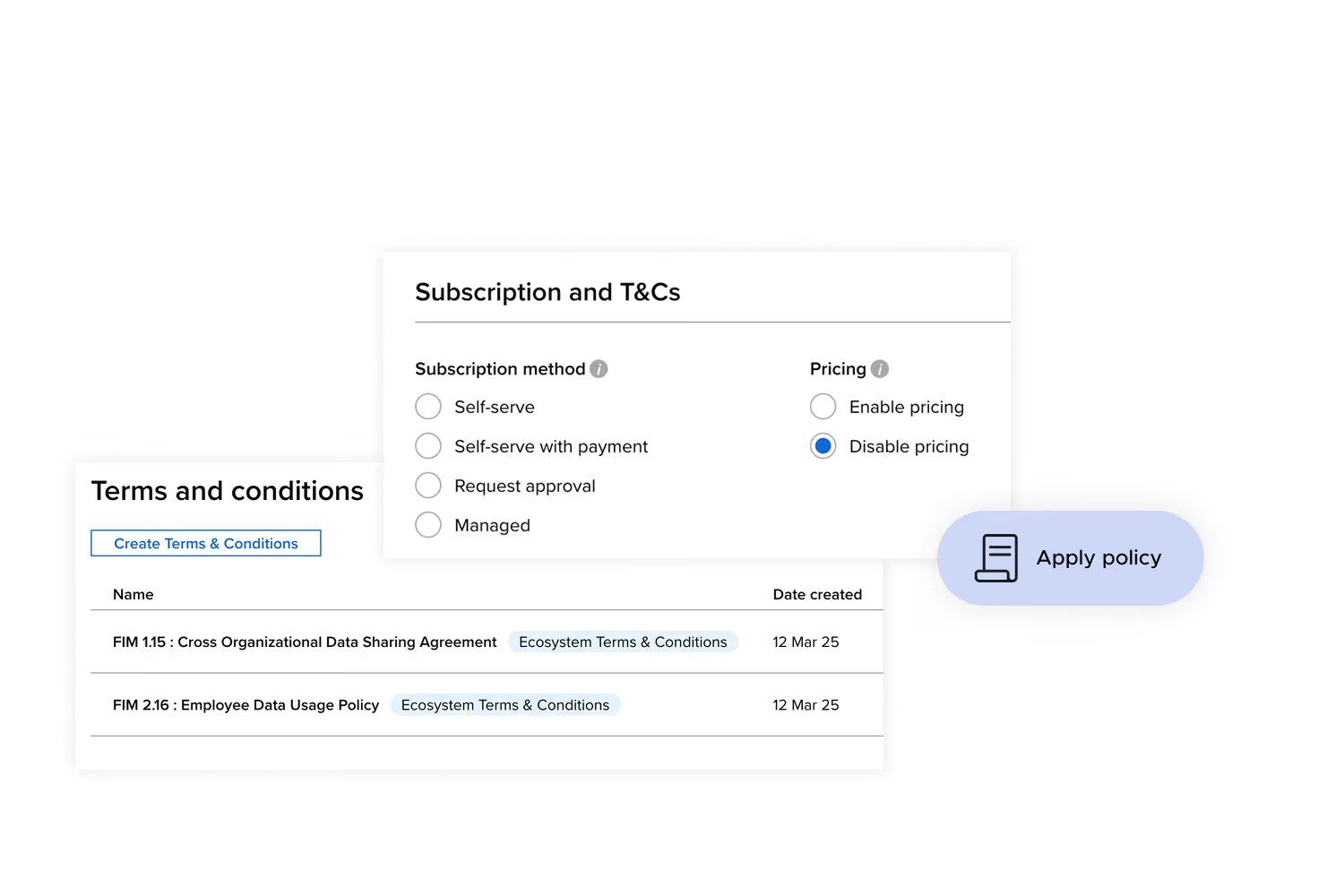
Repeatable governance under scrutiny
Replace manual approvals and ad hoc provisioning with a consistent model designed to satisfy research ethics committees, ICO obligations, MHRA requirements, and commercial data agreements.
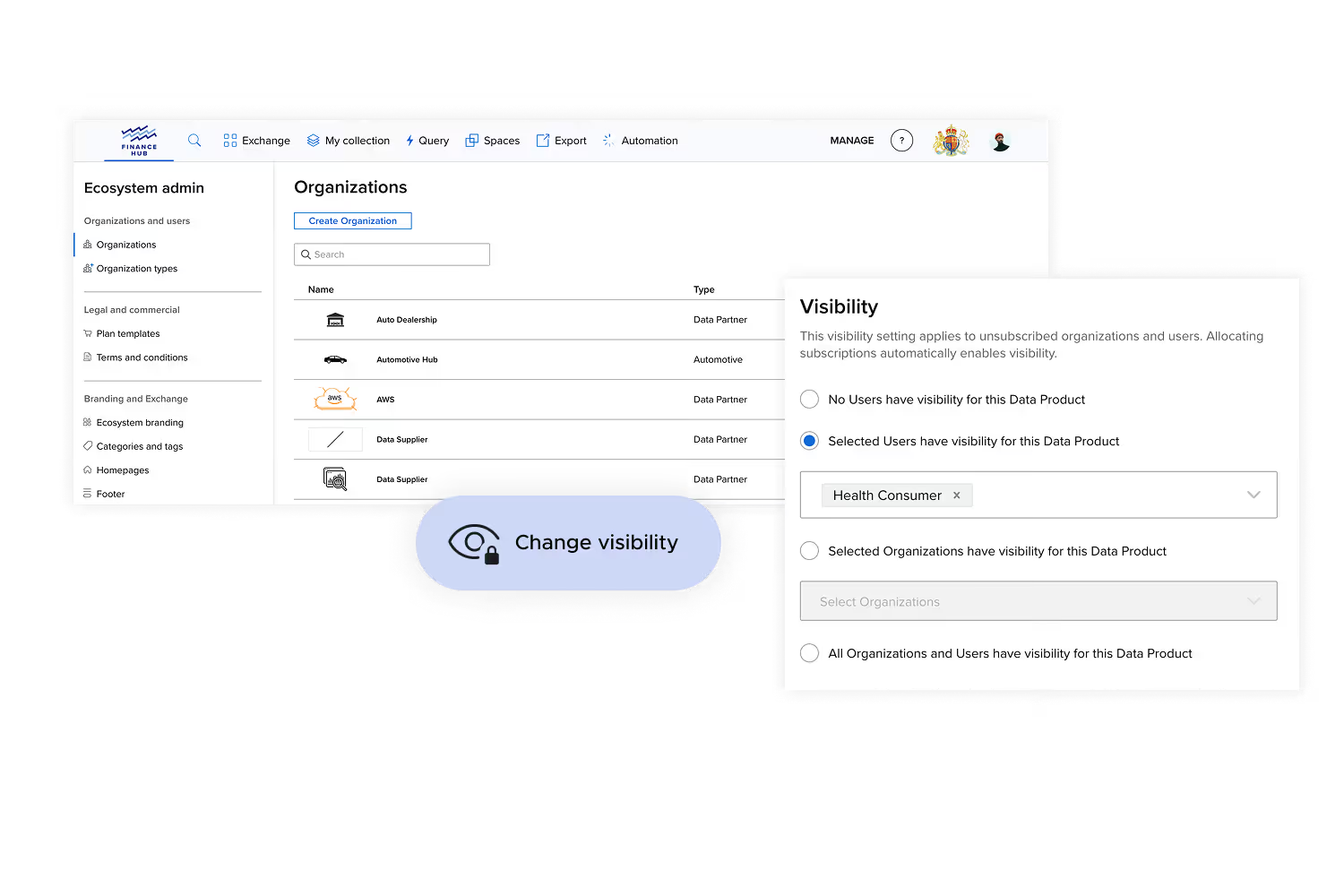
Precise access. Consistently enforced.
Access is governed through entitlements that reflect your policies and agreements. Entitlements are applied consistently across both research environments and data storefront delivery.
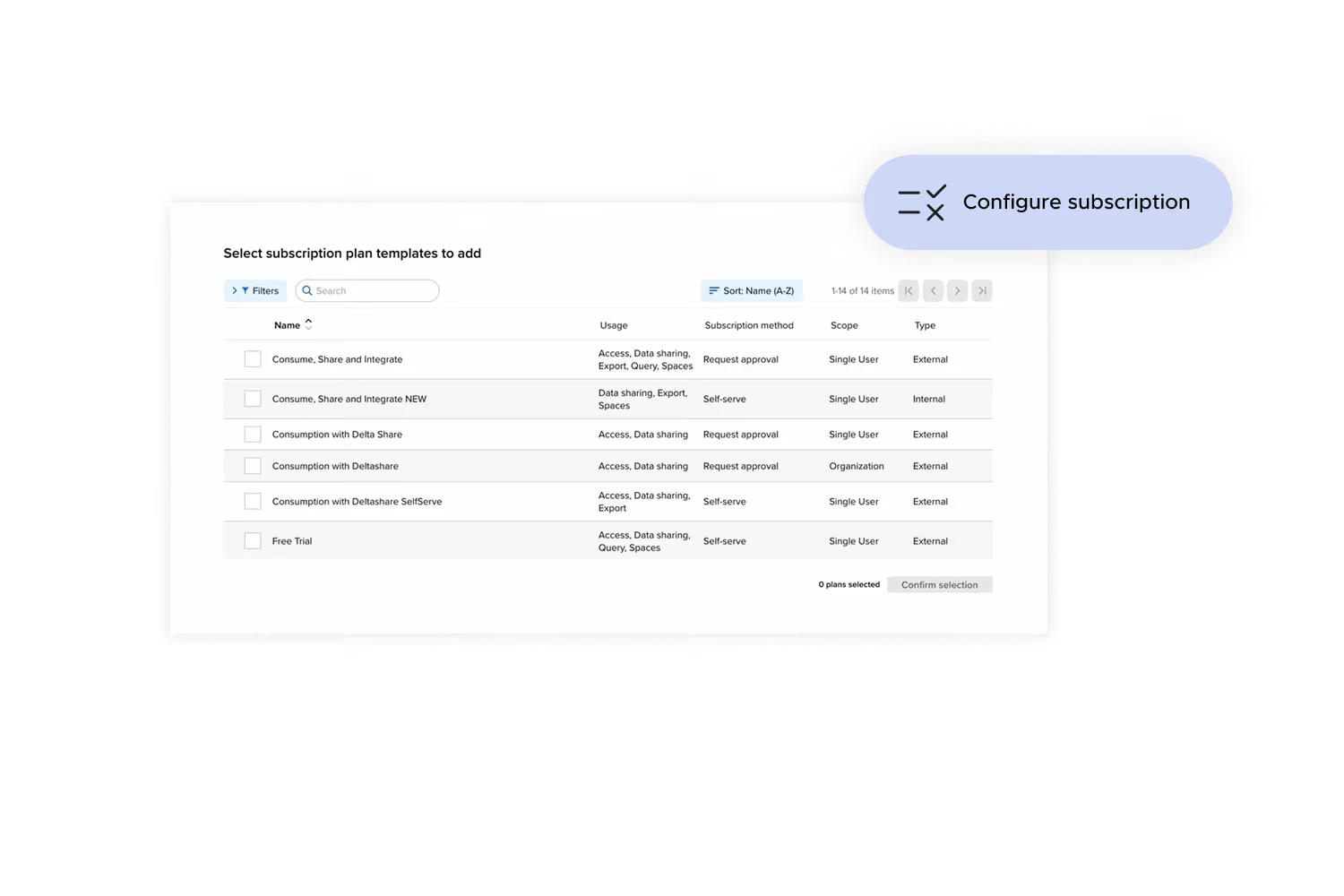
Policy made operational
Access is managed through entitlements that reflect your licensing terms, data sensitivity classifications, and permitted use cases. External parties are provisioned with exactly what they need, while conditions and restrictions are enforced by design.

Evidence that satisfies the most demanding reviewers
Granular logs of who accessed what, when, under which terms, and in which environment. Produced automatically across all external sharing without additional configuration – whether through the research environment or the data storefront.
Built for the teams who own the value — and the risk
Harbr is for the teams who own external research collaboration and data distribution in health and life sciences — and for every compliance and governance stakeholder who needs to know it's under control.

Research data & informatics platforms
Responsible for enabling secure external collaboration on sensitive research data. Harbr removes the need to build bespoke environments project by project — providing a repeatable operating model that shortens time-to-collaboration.

Data governance and compliance
Responsible for ensuring external data access is lawful, auditable, and defensible. Harbr provides a single system of record for who accessed what, under which terms, and in which environment — supporting internal accountability and external regulations.

Data product & commercial teams
Accountable for distributing health data products to research teams, AI systems, and commercial customers. Harbr provides the storefront infrastructure and entitlement model to scale external access without rebuilding delivery workflows per customer.
Common questions
Many organizations do — but these are built for a specific project or partnership. The question is whether those environments apply consistent governance, produce reliable audit evidence, and can be reused for the next collaboration without rebuilding from scratch. Harbr is the platform that makes it possible to build on your success over time.
Existing mechanisms typically handle delivery. Harbr adds the layer above: governed discovery, subscription-based entitlements, self-service evaluation, and complete audit trails — so distribution is commercial, controlled, and scalable, not just functional.
That's exactly the environment Harbr is built for. Data stays at source or within controlled environments where it's analyzed in place. Entitlements are enforced consistently. Audit trails are produced automatically. Harbr doesn't add any governance risk — it reduces it.



